How Caffeine and Vitamin D Genes Influence Bone Health and Osteoporosis

Vitamin D increases the uptake of calcium from the intestine into the blood stream, which the body then uses to mineralize and strengthen bone. Healthy bones are strong but with a surprising degree of flexibility. Osteoporosis is the medical condition that arises when bones become brittle and fragile making them more susceptible to fracture.
This is often associated with a deficiency of vitamin D, or calcium, but also correlates strongly with age, especially in postmenopausal women who are at high risk.
So, knowing that calcium absorption is linked to proper vitamin D activity and that vitamin D binds to the VDR protein to induce this effect, a key question is are any VDR gene polymorphisms associated with bone health?
Several studies have shown that the shorter VDR protein produced by the presence of the VDR rs2228570 (FokI) ‘C’ allele is more “transcriptionally active.” This means that given the same levels of vitamin D, someone with the short ‘C’ allele VDR protein will display increased expression of vitamin D dependent genes. (1, 2) This increased activation of vitamin D dependent genes results in a greater uptake of calcium from the gut, and has shown to increase bone density, which is associated with improved bone health (3, 4).
Caffeine and bone health
There have been numerous observations that an increase in caffeine intake associates with poor bone health and a risk of developing osteoporosis, but a conclusive effect has never been defined. However, with an aging population and our ever-increasing caffeine intakes the area is being more thoroughly investigated, especially in relation to older or elderly women for whom osteoporosis can be a serious issue.
A lot of the studies I discuss below refer to caffeine intake as milligrams (mg); but before we get started I thought it would be a good idea to see just how much caffeine is present in typical hot, soft and energy drinks. As an additional note, whilst there is no firm guidance on upper caffeine intake 400 mg/day is widely accepted to be a safe maximum.
| Drink | Caffeine (mg) |
| Coffee | |
| Filter | 95-200 |
| Decaf Filter | 2-12 |
| Espresso * | 47-75 |
| Decaf Espresso * | 0-15 |
| Instant | 27-173 |
| Decaf Instant | 2-12 |
| Tea | |
| Black | 14-70 |
| Green | 24-45 |
| Iced | 5-40 |
| Soft & Energy Drinks | |
| Coca-Cola | 15-23 |
| Pepsi | 21-26 |
| Dr Pepper | 24-28 |
| Mountain Dew | 28-36 |
| 7UP | 0 |
| Red Bull | 75-80 |
| Amp | 71-74 |
Amount of caffeine in mg present in 8 oz (240 mL) serving. * 1oz (30 mL) Table adapted from Mayo Clinic, Nutrition and healthy eating, Caffeine content for coffee, tea, soda and more.
Drinking caffeine can cause greater excretion of calcium
Now that we know how much caffeine is contained in common drinks, let’s investigate how it is thought to impact on bone health.
Firstly, the diuretic effect of caffeine has been proposed to lead to an increase in urinary calcium excretion and decrease in absorption from the intestine.
Secondly, it has been proposed that caffeine may itself directly inhibit the production of new bone (8, 9, 10). Based on these proposed mechanisms studies have been performed investigating the impact of caffeine on bone health with varying results.
However, a recent meta-analysis of the data available has described a noticeable effect. Relative risk of fracture increased by 4.9% in women overall and conversely, there was a 9% drop in relative risk of bone fracture for men for each cup of coffee per day. Interestingly, they did not observe any age-related effects even in postmenopausal women who had been identified as being at high risk of developing osteoporosis. One important note is that there have been relatively few studies looking at the response of men to caffeine so the authors of the paper suggest that the beneficial effect of caffeine in men may be overstated. But what does this relative risk actually mean?
Well in this instance it refers to the risk of a person who drinks one cup of coffee a day suffering a fracture, compared to someone who drinks no coffee. But without knowing the actual risk this data is meaningless. Luckily the graph below can help with this. As you can see the actual risk of fractures in those below the age of 65 is very low and roughly equal between the sexes. For example, in the 18-44 category about 10 women in 10,000 will suffer a fracture each year. A 4.9% increase in relative risk would raise this to 10.5 women so the effect is tiny. However, if we look at the 75-84 age group we can see that over 200 women per 10,000 will suffer a fracture each year, a 4.9% increase would increase this to 208. So still a small effect, but importantly this is based on drinking a single cup of coffee, with the risk doubling for each additional serving (11). This effect was confirmed by a later study (12). 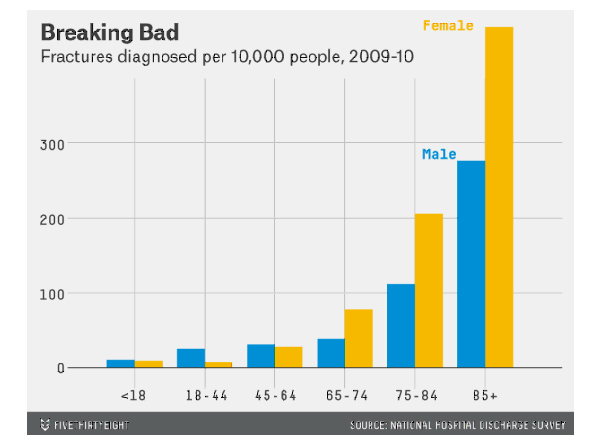
Bringing it all together
Can we link everything together?
We know that VDR gene polymorphisms can effect general bone health. We know that increased caffeine intake is related to a decrease in bone health in women, which has more impact in those already at risk of poor bone health. This study from 2001 by Rapuri et al., showed that postmenopausal women homozygous for the rs731236 (TaqI) ‘C’ allele who ingested over 300mg of caffeine a day showed a 9% decrease in bone density in the spine compared to those with low caffeine intakes, as you can see in the graph below. 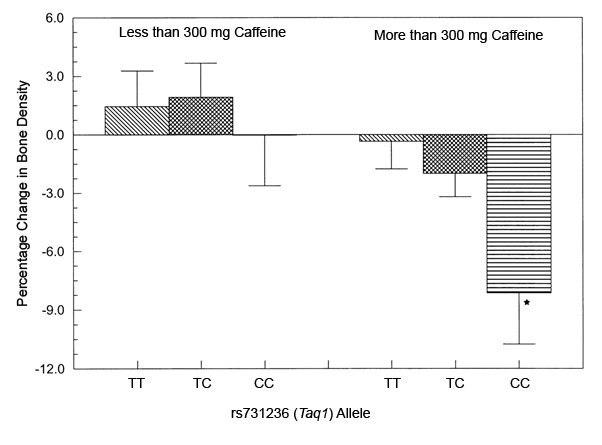
Take-home message
We’ve covered quite a lot here so for a summary I think a list of simple bullet points may be best:
- Vitamin D is important to promote calcium uptake from the gut, which promotes healthy bone formation.
- Variants in some vitamin D genes can negatively affect general bone health;
- High caffeine intake is associated with reduced bone health, particularly in postmenopausal women who are already at high risk.
- A single study investigating VDR gene polymorphisms, caffeine intake and bone health found that high doses of caffeine and the rs731236 ‘C’ allele did correlate with poor bone health but there has been no follow up study to date.



