Why Using Cannabis is Bad for Brain Development in Certain Genotypes
Article at a Glance
- There does appear to be a link between cannabis use, especially before age 25, and mental illness, or psychosis.
- Polymorphisms in the dopamine related genes such as AKT1 and COMT play a role in the likelihood of developing marijuana induced psychosis.
- The higher the THC in a marijuana product, the more dangerous it becomes. Best to avoid synthetic THC and mega doses of THC in favor of products that contain more CBD.
Genes Mentioned

Contents
Cannabis is one of the hottest health topics around right now. It’s been linked with a vast number of positive health effects such as increased appetite in cancer patients and many swear by its ability to help them sleep. It’s clear cannabis has some medicinal properties. However, there is a growing chorus of voices concerned about the mental health implications of marijuana use in children and there does seem to be a link between cannabis use at an early age and increased risk for psychosis. A recent study in rats warns that cannabis use in teens may permanently alter brain development.
When you look at the evidence, our view is that both the risks, and the benefits, of cannabis are currently oversold. Let’s dig into the research.
What is Cannabis?
Firstly what exactly is cannabis? Well, broadly speaking there are three species of cannabis plant, Cannabis sativa, Cannabis indica and Cannabis ruderalis. In social circles, sativas are known as “uppers” and indica strains are described as more “mellow.” Indica strains are popular for people using cannabis for sleep. The cannabis plant is originally thought to have originated from central Asia, but is now grown worldwide for medical, industrial, and recreational use. In fact it’s a common misconception that hemp and cannabis are separate species, but hemp can be made from the stalk of any cannabis species.
How do Cannabinoid Receptors Work?
The psychoactive effect of cannabis is driven by a family of molecules known as cannabinoids (nearly 500 have been described), with tetrahydrocannabinol (THC) being perhaps the most active. CBD, the popular “healthy” ingredient in cannabis is also a cannabinoid, but as we will learn in a moment, it blocks the activity of the cannabinoid receptor, rather than enhancing it as THC does. As a family, cannabinoids act on the cannabinoid receptors (CB1 – CNR1 and CB2 – CNR2) found on cells which control the release of neurotransmitters in the brain. So these receptors already exist in the body and are used to regulate a whole host of processes relating to anxiety, pain, inflammation, gastrointestinal activity and motor control. The receptors, and the factors which act on them in the body, are termed the endocannabinoid system. Upon activation, the receptors are thought to stimulate a complex intracellular signaling pathway, that ultimately results in a reduction in the release of glutamate and GABA. Of the two receptors, CB1 is thought to have the largest effect in the brain as it is expressed more highly. Based on those systems you can see how the cannabinoids expressed in cannabis can lead to a lot of the positive and negative effects associated with the drug, depending on how exactly they interact with the receptor, more detail below when we look at two key cannabinoids with quite different effects.
Does the fact that we have Cannabinoid receptors mean we were designed to use Cannabis?
A common misconception around the naming of the cannabinoid receptors (and the system) is that it means we were designed to use cannabis. The truth is actually rather more prosaic and has to do with how the receptors were discovered. It was long known that cannabis has a profound effect on the brain, but it wasn’t until 1988 when researchers were first able to determine this effect was being modulated at the molecular level. The researchers used a synthetic cannabinoid and were able to identify and isolate a receptor in the brain which responded to cannabinoids, and hence the name was born. It was later discovered, in 1992, that our body produces its own molecules which target these receptors, and so these became known as endocannabinoids, or to break that name down, cannabinoids from within. Had the receptors been discovered without using cannabinoids they would likely have been named something else entirely. So, no, just because we have cannabinoid receptors does not mean we were “designed” to use cannabis.
Tetrahydrocannabinol (THC)
THC is the main psychoactive component of cannabis, and it acts as a “partial agonist” of the CB1 and CB2 receptors, which means it binds and has an effect, but not as strong an effect as “full agonists” such as 2-arachidonoylglycerol. THC activates the cannabinoid receptors, much more so when it is delivered in synthetic forms. Synthetic forms of cannabis, such as spice, have been designed with a much more potent effect accounting for the often severe effects experienced by users. Simply put the receptor is being hit by a high dose of a very potent agonist, for a prolonged period of time, whereas natural cannabinoids may be expressed at a lower dose and only for short time periods. THC from cannabis is much less potent than its synthetic counterparts. Outside of CB1 and CB2 THC has also been shown to bind to other receptors with varying effects, although these are typically less well studied. 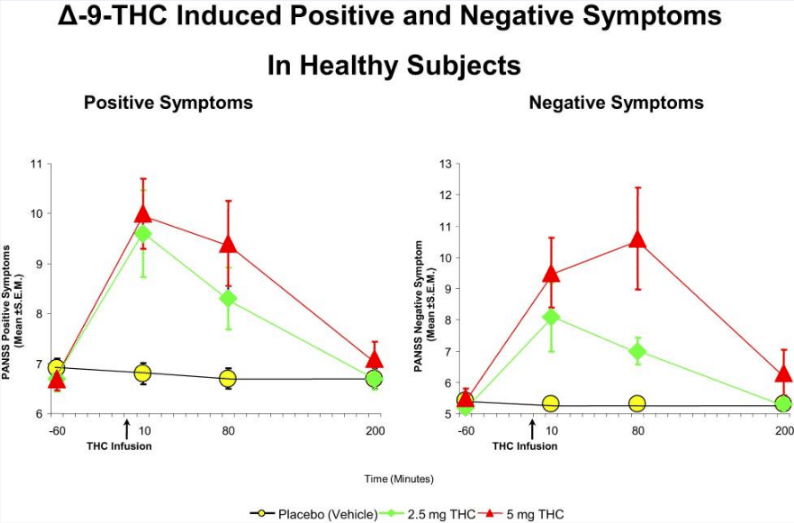
Cannabidiol (CBD) – the Non-psychoactive Cannabinoid
CBD is also found in cannabis but does not have the same psychoactive effects as THC, with some studies suggest that it actually acts to modulate the harmful effects of THC. CBD also works through the CB1 and CB2, but rather than being a partial agonist, it is said to be a “partial antagonist”, meaning it turns off the pathways activated by THC. So, in effect it is opposing the action of THC. Things aren’t quite so simple though as there is some evidence that CBD may also upregulate some of the responses to THC by increasing the number of CB1 receptors present. So, bottom line is that CBD can make the impact of THC stronger when high THC products are used. Furthermore, CBD as with THC has also been shown to bind to a wide variety of other receptors, which are all thought to drive its antipsychotic effects, including the serotonin 1A receptor and delta opiod receptor. if THC is a pro psychotic, CBD has the exact opposite effect.
Studies on Cannabis Induced Psychosis
So onto the main focus of the article. Psychosis is something we’ve all probably heard of but the definition can actually be quite nebulous. For a working definition, two main features are:
- hallucinations where a person hears, sees and, in some cases, feels, smells or tastes things that aren’t there, a common example being hearing voices, and
- delusions where a person has strong beliefs that aren’t shared by others; a common delusion is someone believing there is a conspiracy to harm them.
Psychosis can be caused by underlying issues such as depression or schizophrenia, or it can be triggered by more acute issues such as a traumatic experience, stress or as a response to drugs (a whole host not just cannabis) and alcohol.
Mechanisms
So as you can see, psychosis is a catch-all diagnosis, which is why it’s difficult to investigate. Diagnosis is tricky, and making a consistent diagnosis that works for large groups of people is trickier still. The field of measuring such events is known as psychophysiology, or more simply attempting to observe psychological effects by measuring physiological metrics. Typically this refers to the use of electroencephalography (EEG) to map what is happening in the brain following various stimuli. One stimulus that is often used is termed the P50 response. The P50 response is an event which occurs in the brain approximately 50 ms after being presented with a simple stimulus, like a click or a beep. Patients with schizophrenia have been shown to have an abnormal suppression of the P50 response, and studies comparing chronic cannabis users to non-users showed a similar suppressed P50 response, even after abstaining from drug use for 24 hours, in a dose-dependent manner (the more you smoke the bigger the effect). Another measure is the P300 response, which is typically measured when an individual is presented with a stimulus and is asked to decide on a response. As above, deficits in P300 have been demonstrated in patients with schizophrenia 2 3 4. Further studies then showed that THC caused a similar deficit in response to occur in healthy individuals 5 6. However, studies in long-term cannabis users are more confusing with both positive and negative effects described 7 8. The opposite effect was observed when looking at mismatch negativity, a response that occurs in the brain after hearing a deviant auditory stimulus in a sequence of normal stimuli (so, for example, imagine someone saying S over and over then occasionally inserting a T). Deficits in MNN have been shown in people with schizophrenia and early psychosis 9, and also in long-term cannabis users 10. However, studies attempting to replicate this in the lab using THC did not see any effect.
Epidemiology
The first study linking cannabis with psychosis was published in the 80’s and investigated the association between cannabis and psychosis in a population of 45,000 Swedish conscripts. The take-home message from the study was that conscripts who reported having used cannabis at least once in their lifetime had a 2.4-fold increased risk of developing schizophrenia over the course of 15 years and this relative risk rose to 6-fold in those who had used cannabis more than 50 times in their lifetime 11 12. However, several issues have been raised with the study over the years, the first is to do with directionality, as in are those who smoke cannabis more likely to develop schizophrenia, or are those who are more likely to develop schizophrenia more likely to smoke cannabis? This is an issue which dogs many of these studies and is a difficult one to address when a clear mechanism remains unknown 13. Other issues relate to the difficulty in controlling for other factors such as other drug use, and other factors which may arise from the fact that such studies are self-reporting.
| Consumption | Number (Percentage) | Cases of schizophrenia | Relative risk | Confidence Intervals |
|---|---|---|---|---|
| 0 | 41,280 (90) | 197 | 1.0 | - |
| 1-10 | 2,836 (6.2) | 18 | 1.3 | 0.8 - 2.2 |
| 11-50 | 702 (1.5) | 10 | 3.0 | 1.6 - 5.5 |
| >50 | 752 (1.7) | 21 | 6.0 | 4.0 - 8.9 |
THC and CBD Dosing
A major issue with a lot of these studies is the use of THC at defined doses, rather than the cannabis that your average person is likely to use, which may have a lower THC dose, and also contain CBD which is negating some of its activity. This is likely one cause of the difference between the well-controlled lab studies, which show clear psychophysiological effects, which then become much messier when applied to people in the real world (this is not to discount those studies but shows there are multiple factors at play). As discussed above CBD has antipsychotic properties, and so this effect is likely missed in these studies, which rely purely on THC. Similarly for epidemiological studies, the type of cannabis consumed, and therefore the amount of THC and CBD contained within is typically not controlled for. This issue is likely in part to the often illegal nature of cannabis, making strain assessment and reporting in these types of study difficulty. Hopefully, with a loosening of cannabis laws (and also a standardization/better description of legal cannabis products) this may be addressed. This issue is especially important as the THC content in cannabis has increased dramatically, with an increase from around 3.5% in 1993 to 8.8% in 2008 22.
Avoid High THC Pot
High THC products, especially synthetic ones, seem to have greater potential for harm, but CBD seems well tolerated at a variety of doses. So the take home is avoid high THC, rather than go for high CBD to balance high THC.
Factors to Consider
So it’s not a simple cut and dry case, as with every complex part of our health (and mental health is extremely complex) there are lots of factors that modulate the response.
Age of exposure
Age of exposure is widely recognized as being the most important driver, with the earlier the age of exposure to cannabis, the greater the risk of a psychosis outcome 23. Both the Dunedin and CHDS studies described above support this finding, with a subsequent large meta-analysis of 80+ studies demonstrating that the age of onset of psychosis in cannabis users was 2.7 years younger than in non-users 24. The mechanisms surrounding this are unclear, as it’s a difficult effect to measure ethically in adults, let alone children. During adolescence and young adulthood brain development continues at a pace, and the brain is incredibly plastic, therefore any factors which interfere with this process can have lasting effects. The endocannabinoid system plays an important role in several processes important in neurodevelopment, and as such disruption of the endocannabinoid system by excessive or non-physiological stimulation, as may be the case with exposure to exogenous cannabinoids, may have far reaching consequences 25 26 27 28. There is some debate about these findings, and their relevance, for example teenagers who begin cannabis use at an earlier age may represent a group that differs in ways that have not been accounted for (such as history of abuse or family socio-economic level) in the aforementioned studies.
Family history of disease
Some studies have indicated that a positive family history of schizophrenia may increase risk for cannabis-induced psychotic disorders. For example in a retrospective study, those patients admitted for acute psychosis, who tested positive for cananabinoids in their urine, were 10 times more likely to have a family history of schizophrenia 29, with later studies confirming this effect 30 31. So with a strong familial link, there must be some genetic factors which are correlated?
Genetic factors
So outside of strain variations and environmental factors is there anything in our genes that controls our response? Two main SNPs have been linked with the development of psychosis, one in the COMT gene, and one in ATK1. Some weaker or inconsistent effects have also been described for SNPs in the DAT1, NRG1 and CNR1 genes.
COMT – rs4680
COMT was the first genes linked with development of psychosis and the use of cannabis, in particular in teenage populations 32. The mechanism is sound as COMT plays a critical role in the breakdown of dopmaine, with the rs460 SNP being linked with a 40% reduction in enzyme activity in those with the ‘A’ allele. The 2005 study by Caspi et al, showed a 10-fold increase in the risk of developing psychosis in those with the ‘G’ allele, with homozygotes fairing the worse. This effect was validated in a controlled trail where those carrying a ‘G’ allele were more likely to develop psychosis following exposure to THC 33. However, subsequent studies have painted a less clear picture with several studies not finding any association 34 35 36, and other studies only finding an effect when associated with other environmental factors such as a history of childhood abuse 37 38
ATK1 – rs2494732
In those who regularly smoke cannabis there was a significant increase in the risk of developing psychosis, especially in those carrying two copies of the risk ‘C’ allele where a seven fold increase in risk was reported. AKT1 is known to respond to dopamine 39, and THC has been shown to induce dopamine release 40, and also activate AKT1 through the CB1 and CB2 receptors. The authors hypothesise that the ‘C’ allele of of AKT1 leads a reduced responsiveness to dopamine. Therefore when cannabis is used, THC stimulates dopamine release. This dopamine remains active for longer periods in those carrying the risk ‘C’ allele, as it is not processed as efficiently. Importantly, dopamine remaining active for long periods without processing has been associated with the development of psychotic symptoms 41.
Wrapping up
To wrap up, there is consistent evidence of an association between cannabis use and the development of psychosis, although this is modulated by a whole host of other factors making it difficult to assign a total risk. Those with a family history of psychosis should strongly consider avoiding cannabis, and exposure should definitely be limited to adult populations. At the genetic level there is some clear evidence for the involvement of SNPs in ATK1 and COMT and carriers may wish to consider their use of cannabis. An important step for everyone, would be to avoid synthetic forms of THC entirely if you want to then use a cannabis strain with a lower THC content, and this step should hopefully become easier as cannabis is legalized in more states and countries. CBD as a supplement has shown a high level of tolerance with low levels of adverse reactions, but as with any supplementation, the best way is to start small and work up to a higher dose (within recommendations). As ever, happy to hear thoughts and inputs as recognize that this is quite an interesting topic, so hearing your feedback would be great.



I suffer from ankylosis spondyloitis, fibromyalgia. I’ve been an RN for 25 years. Article is so true about marijuana and acute psychosis. I had it happen to me, what a horrible experience. My pain mgmt doctor asked if l was interested in trying medical marijuana. One because l wanted off opiates(7 years) and wanted something closer to the Earth (not pharmaceuticals). I started CBD oil and vape pen with MM. Did not like the way it MM made me feel. After about four days l was having problems sleeping and some paranoia. At that point, l became sleep deprived. For three days l was in my house functioning, not knowing what I was doing. Can’t recall any of the days. My husband called my daughter because l was acting strange and fighting with him(not a normal activity). I was counting pills, talking to invisible people, called my son told him there was a mob of people on my lawn wrapping themselves in blue carpets. That they wanted me to kill myself. My daughter didn’t know what to do. She called police for help. I remember the police being in my bedroom. Telling my daughter go home and I’ll sleep then kill myself later. Also remember being handcuffed in back of police car, bless that officer, l could tell he didn’t like putting the cuffs on me(it’s protocol).I do have some depression, being l can no longer work and l loved my job, so rewarding. My pain management doctor was beside himself with what happened. I thought maybe the weaning off opiates, but l was already on week three. There were no answers we were both dumbfounded. I was in a lock down psych floor for two days. Horrible experience, now l understand why and how psych pts fall between the cracks. Not one nurse on all shifts showed any compassionate towards me or other patients. I sat there for two days, being ignored, watching everything going on (sure l missed somethings on arrival). Only compassionate person was a CNA he actually talked to me.Did sleep the first day and second day after being up l fell asleep at 2am, they woke me at 5am to tell me l was discharged. So no one gave me a answer to why l was there. I did see my primary after discharge she asked what happened, l had no answer. I saw one doctor on admission pretty sure diagnosed with depression, and l know l am. No psychosis in family, some anxiety in family. 5 days later l received my DNA results from Prometheus and ta da my answer: l have the AKT1gene, rs2494732 putting me at greater odds of cannabis-associated psychosis. Made sense, that not vaping for two days my symptoms went away. So beware of that cannabis, not for everyone. I continue on CBD oil, excercise using rebound classes, stopped eating processed foods, eating organic food and meat, low inflammatory foods. Haven’t felt this good in years. Glad I’m off opiates. I did continue using vape pen only at bed time and very small puff, also took Advil for discomfort. After l received my report l stopped MM altogether.
Good connections Dr Gardner. Appreciate also weighing in on genetic influences of CBD on CYP2C19, effect on arrhythmias, cognitive executive functioning as most data refer to cannabis not CBD. Thanks