MTHFR Health Impact Commentary Tends to Ignore the Black Community
Article at a Glance
- The impact of common MTHFR mutations is often overstated by holistic health practitioners.
- While not as impactful as many claim, especially homozygous MTHFR mutations can impact heart health.
- Further, a 2016 meta-analyses (a large study combining the results of many smaller studies) of 47 studies published on the subject of MTHFR contained only one study evaluating an African population.
- T the T allele of MTHFR C677T is an important marker for the Black Community in particular to test for, as even one copy has been shown to increase heart attack risk.
Genes Mentioned

Contents
If you’re interested in personalized nutrition, you will have undoubtedly come across the protein methylenetetrahydrofolate reductase (MTHFR).
MTHFR is an enzyme which is encoded by the MTHFR gene and functions to convert 5,10-methylenetetrahydrofolate (MeTHF) to 5-methlytetrahydrofolate (MTHF).
All people have MTHFR genes, and the polymorphisms (“mutations”) are very common, although prevalence varies by ethnicity.
23andme on MTHFR: no health impact
Many of the health claims surrounding MTHFR range from overstated to outright false. This has led some in the scientific community to dismiss MTHFR as “not worth testing for,” with 23andme concluding that changes in the MTHFR gene have next to zero health impact.
However, just as overstated claims about MTHFR are not consistent with the weight of the scientific evidence, the total dismissal of MTHFR as having no health impact is simply not true, especially for the Black community.
If there was ever a case of throwing the scientific baby out with the bath water, MTHFR is that case. The presence of an MTHFR variant does not justify skipping vaccines, and no, the presence of MTHFR does not mean a baby will be born with autism. However, in a rush to debunk false claims about MTHFR, we are ignoring important research about cardiovascular health, research we hope to bring to light in this blog post.
Here are the facts:
- Heart disease is the leading cause of death in this country
- From age 18-49, the Black community is twice as likely to die from heart disease as is the White community
- Black Americans ages 35-64 are 50% more likely to suffer from high blood pressure than are White Americans
- Black Americans who carry the T allele for MTHFR C677T have a 63% increased risk for heart attack
What sounds like a pretty un-inspiring aspect of B vitamin metabolism has actually become one of, if not the, most investigated reactions in the field of personalized genetic medicine, but the public does not have the time to review this research themselves. Until something changes, and more of us speak out, MTHFR Google searches return a consistent message – any website or physician discussing MTHFR is engaged in pseudo-science.
MTHFR symptoms and conditions
This message ignores important research from major medical journals demonstrating an association with MTHFR variants and several disorders including heart disease, 1 stroke, 2 high blood pressure, 2 various neurological issues 3 and potential issues with birth defects. 4
I am going to dive into the science of the MTHFR gene and offer an overview of the related biochemistry, but before I do, I want to point out that different ethnicities may have different health outcomes when this polymorphism is present.
One of the major challenges facing the researchers conducting clinical trials is inclusion. The sad reality is that so many of the studies we see are conducted only in White populations. Consider this 2016 meta-analyses (a large study combining the results of many smaller studies) of 47 studies published in the International Journal of Cardiology. If you read just the top level findings, the 23andme approach to MTHFR research appears to be confirmed.
The meta-analyses begins with this:
A total of 47 studies were finally included in this meta-analysis. Overall, the results showed no statistically significant association between C667T and A1298C polymorphisms and MI risk.
Open and shut case, right?
Just another study dismissing the science on MTHFR. Not so fast. When you continue reading, you find something quite remarkable – the meta-analyses found a 63% increased risk for heart attacks in Black and African populations who carry a copy of MTHFR C677T!
To quote the meta-analyses:
in subgroup analysis by ethnicity, the T allele of C677T polymorphism was associated with a 63% increased risk of MI compared with the C allele in African populations.
Further, of 47 case-control studies regarding the C667T polymorphism in the meta-analyses, there was only a single study involving an African population.
23andme’s objection to MTHFR “hype” is predicated on the idea that we can’t glean health impact from a single SNP. As a general rule, we agree.
However, the finding that Black people have a 63% increased risk of heart attack based on a single genetic marker is significant. We note that 23andme does not include the meta-analyses from the International Journal of Cardiology in its MTHFR blog post citations, and neither do blog posts like this one from Self Magazine which confidently tell people not to even test for MTHFR variants. Don’t test for a marker that is associated with a 63% increased risk for heart disease in a population already at greater risk for heart disease?
This doesn’t seem like good advice.
Unfortunately, many of the studies that we have on MTHFR have looked only at Caucasian populations, with mixed evidence. However, there appears to be greater evidence of health impact for the Black community, especially as it relates to heart disease. It should be pointed out that the occurrence of the 677 C > T variants is less often seen in Black populations than in White and Latinx populations, however, the fact remains that we must have a more nuanced and more inclusive conversation about the health impact of MTHFR.
Now to the drier part of this piece, a run down of the basic science behind this much discussed, and debated, gene.
MTHFR and the one carbon pathway
MTHFR actually sits in the middle of a rather complicated pathway, called the one carbon pathway, which is involved in the conversion of dietary folate into a variety of other products which are key in the synthesis of DNA, RNA and amino acids, so it’s pretty fundamental to life.
Two “famous” polymorphisms in MTHFR have been identified which lead to a reduction in enzyme activity; rs1801133 (C677T) and rs1801131 (A1298C) which are detailed in the table below.
| Protein | Gene | SNP | Major Allele/Minor Allele (Risk) | Risk |
|---|---|---|---|---|
| MTHFR | MTHFR | rs1801133 (C677T) | C/T | C (Ala) > T (Val) leads to a heat-sensitive enzyme with reduced activity. Also the required co-factor vitamin B2 is released more quickly by those with the ‘T’ allele [R]. |
| rs1801131 (A1298C) | A/C | A (Glu) > C (Ala) leads to a smaller reduction of enzyme activity than described above through an unknown mechanism [R]. |
How can MTHFR SNPs lead to poor health?
As discussed in the table above, the those carrying the ‘T’ allele of the rs1801133 SNP display a reduced MTHFR enzyme activity of between 30-70%; which leads to a reduction in the conversion of 5,10-MeTHF to 5-MTHF. The Black community seems to see a disproportionate impact with these polymorphisms and heart health.
It is thought that this occurs because the enzyme is rendered more heat sensitive by the substitution of a valine for an alanine, and also has a reduced ability to stay bound with its co-factor, vitamin B2. 5 The ‘C’ allele of rs1801131 also reduces the enzyme activity of MTHFR although to a lesser extent, and the mechanism remains unknown. 6
This lack of enzyme activity doesn’t directly lead to the development of the disorders listed above in the MTHFR symptoms section of this post. Rather, it is how the lack of 5-MTHF impacts on the processing of another molecule called homocysteine, which when present at elevated levels, can be very damaging to tissue, and is associated with increased cardiovascular risks, various neurological issues and poor bone health amongst others. 7
Homocysteine is typically converted into methionine, or cysteine, both of which are less harmful. The major driver of this conversion is an enzyme called methionine synthase, although there are other pathways involved as well. Methionine synthase generates methionine from homocysteine using 5-MTHF as a donor for a methyl group. With a lack of 5-MTHF, as occurs in those with less functional MTHFR, this reaction stalls and leads to an accumulation of homocysteine.
The interaction between the one carbon pathway and the processing of homocysteine into less harmful products is quite confusing so I’ve made the diagram below to highlight the key enzymes involved.
The importance of B vitamins
One very interesting thing to note is the importance of B vitamins in both the one carbon pathway and homocysteine clearance. Whilst those eating a healthy, balanced diet may receive a sufficient dietary intake of B vitamins, many of us don’t. So attempts to deal with MTHFR enzyme deficiency by supplementing with folate alone may not prove successful if the enzymes of the various pathways cannot function.
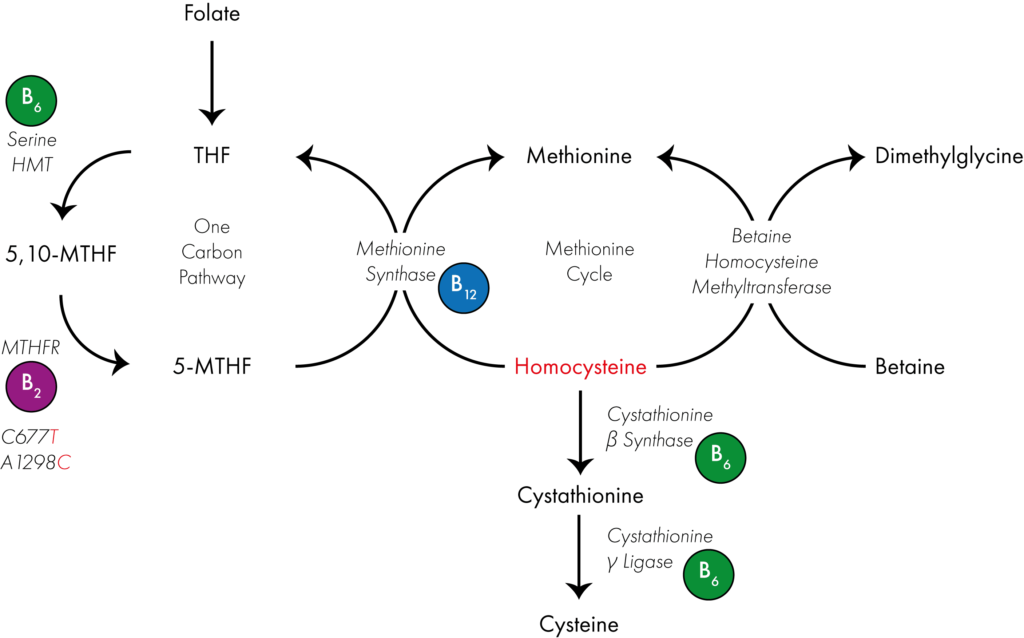
Very briefly dietary folate is converted into tetrahydrofolate (THF) as an entrance point into the one carbon cycle. Serine hydroxymethyltransferase converts THF into 5,10-methylenetetrahydrofolate (5,10-MTHF) using vitamin B6 as a co-factor. 5,10-MTHF is then converted into 5-MTHF via the enzyme methylenetetrahydrofolate reductase (MTHFR), which uses vitamin B2 as a co-factor. Finally methionine synthase, which uses vitamin B12 as a co-factor uses 5-MTHF as a methyl donor to convert homocysteine into methionine, resulting in the reformation of THF.
Simultaneously the enyzme betaine homocysteine methyltransferase uses betaine as a methyl donor to convert homocysteine into methionine. Whilst two enzymes which use vitamin B6 as a co-factor convert homocysteine into cysteine. A block in the one carbon pathway as occurs in those with reduced MTHFR activity can therefore lead to an accumulation of homocysteine.
Are MTHFR SNPs really all that bad?
Not always, no. These are common polymorphisms in some populations. As we point out in our Guide to Nutrigenomics, 30% of the general population carries at least one copy of MTHFR C677T. It is estimated that this number is closer to 10% for the Black population.
As numerous previous studies had proved inconclusive when assessing the role of MTHFR rs1801133 (C677T) and rs1801131 (A1298C) SNPs in relation to heart disease, the large scale meta-analysis conducted by the International Journal of Cardiology was designed to answer some lingering questions.
As we already pointed out, no general association between MTHFR SNPs and increased cardiac risk was observed. However, for several subgroups namely African, North American and elderly populations a significant association was observed, with those carrying the ‘T‘ polymorphism of rs1801133 being at elevated risk of suffering a heart attack.
This is why personalized nutrition is so important. People do not react in a uniform way to genetic changes and have different dietary needs. In the case of MTHFR C677T, this is especially true for the Black community. Parroting the party line that MTHFR polymorphisms have “no health impact” puts historically underrepresented communities at a disadvantage by ignoring information that could help them reduce their risk for heart disease with simple dietary and testing interventions.
MTHFR C677T was also observed to play a role when looking at stroke patients. There was a strong association between the ‘T‘ allele of rs1801133 and the occurrence of strokes.
The rest of the claims made about MTHFR, many of which are addressed in this blog post, have much weaker evidence. As far as I’m aware, robust meta analyses for the other disorders associated with MTHFR mutations are not available, although I will update this post as and when I find any.
What to do if you carry an MTHFR SNP
Well, first things first is a blood test to determine if you have high homocysteine levels.
If detected, supplementation with vitamins B2, B6, B12, methylfolate and betaine may be beneficial, but consult your physician first as all supplements can come with side effects. Additionally, as deficiency in these vitamins is also associated with numerous other disorders, due to their important role as co-factors for numerous enzymes, general supplementation may be beneficial.
It is difficult to find studies investigating the effect of supplementation on issues such as heart health due to the timescales involved and issues with the ethics of clinical trials. However, the effect of folate supplementation for expectant mothers has been widely investigated and has been linked with a reduction in birth defects [13]. The effect was so strong that in the USA and many other Western countries grains and cereals are now regularly fortified with additional folate, usually in the form of folic acid.
This fortification sounds great on paper, however some researchers now think that it may be causing health issues of its own, especially in those with reduced MTHFR enzyme activity [14,15].

Let’s look at this diagram from above again.
In a person with fully active MTHFR enzyme, the One Carbon Pathway will cycle normally. But in someone carrying either of the SNPs MTHFR rs1801133 (C677T) or rs1801131 (A1298C) this activity is reduced. As I discuss above this may lead to a reduction in 5-MTHF and the accumulation of homocysteine, but also importantly the product which MTHFR should be breaking down will also build up as well. If we add an excess of folate into this system through supplementation it is easy to see how this precursor product could rapidly accumulate.
As such just increasing folate intake may not be the best process, rather taking a holistic approach and activating all parts of the pathway to encourage proper cycling may be the way forward. Vitamin B2 is an important MTHFR co-factor.
Take-home message
I hope the above post has helped you understand how the MTHFR enzyme functions and how common SNPs can lead to a reduction in its activity.
Large scale meta-analyses have demonstrated a very significant increased risk for the Black community when copies of C677T are present. Sadly, the large scale review of studies conducted by the International Journal of Cardiology has been ignored in the internet discussions surrounding MTHFR.
We hope this blog post gains attention and helps to foster a more inclusive conversation surrounding a genetic variant that is strongly associated with heart attack risk in communities that have long been underrepresented in the clinical research. In our view, it is quite remarkable that a 63% increased for heart attack, as well as associations with stroke risk, is buried under headline after headline that tells readers the MTHFR gene has “no health impact.” This simply isn’t true for the Black community. Glossing over nuance in research in this manner is exactly the type of scenario Ibram X. Kendi and his crusade for anti-racism is trying to combat.
Do keep in mind that although the effect of supplementation has been widely investigated in expectant mothers, the impact on other disorders, especially in those carrying either MTHFR rs1801133 (C677T) or rs1801131 (A1298C) has not. We want to emphasize again that no supplements should be taken without medical supervision.
However, a rapid, easy test for elevated homocysteine is available for those who are concerned, or those who know they carry one of the above SNPs. If levels are found to be elevated a holistic supplement containing methlyfolate and numerous B vitamins may be preferred to folate alone.
It is our opinion that MTHFR should be tested for, especially in the Black community. The scientific community must do more to understand heart disease, our nation’s number one killer, and why it takes the lives of certain ethnic groups at a rate higher than others.




Very interesting article as “Black” Americans are #1 for Heart Disease which is essentially tied to your diet. Its disheartening to learn the MTHFR research is touted as pseudoscience when right on the surface you can see correlations and parallels between populations who have prevalence of certain diseases. I hope to see more individualized care based on ones specific genetic make-up as we learn that eating a diet based on what your individual body needs and not years of research or testing based off of European people as the standard. In the article it mentions “Black and African” what is the distinction being made? Is that meant to refer to a “black” American in comparison to an African?
Najima,thanks for the thoughtful comment. the “Black and African” refers to studies done in African Americans and populations in Africa. We want to be looking for both so our readers get the full picture about genes like MTHFR which have almost exclusively been studies and reported on in Caucasian populations.
Thankful for this post. We are researching my niece’s MTHFR gene. She is African American, has a complex history, is compound heterozygous; C667T and A1298C. Doctors remain baffled. If the scientific community can do more testing and studies for African Americans, we’d have more relevant answers to our issues.
Thanks for reading Shatoya, and thank you for your comment. We definitely need more research on MTHFR variants in the Black community. The one size fits all click bait articles that are commonplace in search results aren’t helping. Best of luck to you and your family.