Have High LDL-C? Consider These Dietary Changes
Genes Mentioned

Contents
Do you have high LDL-C?
Statin therapy is often the first line of defense, however, there are dietary changes that can improve LDL-C levels as well.
LDL causes heart disease
The causal role of LDL in heart disease risk has been long established, however, the evidence continues to mount. In the last year, the European Atherosclerosis Society Consensus (“EAS”) Panel published a position paper intended to better elucidate the mechanism behind LDL and the progression of heart disease.
LDL-C is labeled as the “bad cholesterol” because these particles have the potential to deposit cholesterol into the artery wall, where the accumulated plaque invites inflammation, eventually causing a heart attack or stroke.
To quote the EAS:
Low-density lipoprotein…is the key deliverer of cholesterol to the artery wall.
Cholesterol that makes its way to the artery wall (and that gets stuck there) causes plaque to form in the chambers of the heart. Plaque in your arteries is evidence that the long process of heart disease has begun. The calcium score is an imaging tool which can measure the amount of plaque that has built up in the artery.
While tests like LDL-C are predictive in nature (higher LDL-C correlates with increased risk of heart disease), the calcium score shows the damage that has already been done.
The foods we eat impact LDL-C levels
Perhaps you have just gotten a blood test back and see high levels of LDL-C. You are concerned and want to know why this has happened.
While none of these reasons are mutually exclusive, at a foundational level, there are generally three reasons you could have high LDL-C numbers:
- You are absorbing a lot of cholesterol from the foods you eat and from your digestive tract;
- Your body is making excess cholesterol in response to a high fat diet;
- You have an impaired ability to clear cholesterol from your system
Cholesterol absorption and LDL-C
In most people, cholesterol levels stay tightly regulated. This means that even if you absorb cholesterol from the foods you eat, your body will make less of it. However, this is not true for all of us. In some people, often due to genetic variants, more cholesterol is absorbed from food, resulting in higher LDL-C.
For example, ApoE4 carriers are more likely to be cholesterol hyperabsorbers. 1
How to determine if you are a hyper-absorber of cholesterol
To determine whether you are a hyper-absorber of cholesterol, doctors will measure for the following phytosterols in the blood:
- Sitosterol
- Camposterol
- Cholestanol
Since the human body doesn’t make phytosterols, when they show up in the blood it means they are being absorbed. The logic follows that if an individual is absorbing heightened levels of plant sterols, they are also absorbing an excess of cholesterol. Individuals with genetic changes in the ABCG5 and ABCG8 genes tend to absorb more plant sterol, with the most extreme cases diagnosed as sitosterolemia.
Boston Heart Diagnostics, an advanced heart lab in Boston, offers its Cholesterol Balance Test, which tests for plant sterols as a sign of cholesterol absorption.
The pharmaceutical drug Ezetimibe, or Zetia for short, works by blocking the absorption of cholesterol. When statins don’t lower LDL-C enough, Zetia is commonly prescribed, and has shown promise in clinical trials for reducing the incidence of cardiovascular events.
For more on Zetia, see my blog post on Zetia.
Saturated fat and LDL-C
Every cell in the body makes cholesterol, but it’s the liver that is the big cholesterol producing engine. Under normal circumstances, the liver simply makes less cholesterol when we absorb more dietary cholesterol from food. However, this is not always the case.
In some people, eating a high fat diet, especially a diet high in saturated fat, causes the body to make more cholesterol. The stepped up production of cholesterol then results in higher LDL-C. In some circles, people that make more cholesterol on high fat diets, and therefore see very high LDL-C, are known as “lean mass hyper responders.”
How to tell if you are a hyper-responder to saturated fat
To determine whether you are making (synthesizing) large amounts of cholesterol, such that your LDL-C is trending too high, physicians test for desmosterol, the precursor in the body to cholesterol production. Desmosterol is also part of the Boston Heart Cholesterol Balance Test.
Elevated desmosterol means you have high LDL-C due, at least in part, to greater production of cholesterol.
This greater production could be the result of an increase in consumption of saturated fat rich foods, for example going on a ketogenic diet.
Eating saturated fat increases LDL-C levels in all people, the question is only by how much.
Cholesterol clearance and LDL-C
Studies show that an increase in saturated fat intake does not significantly alter LDL-C in all of the population. In fact, there is tremendous variability between individuals when they adopt high fat diets.
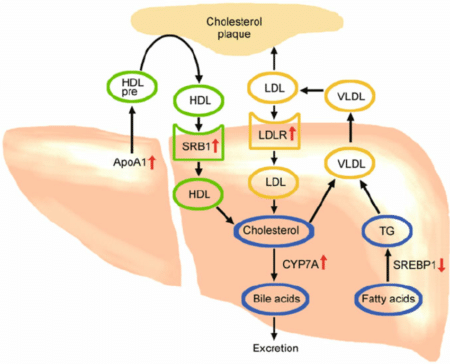
So if production isn’t a major issue for most people, what about LDL removal? This is where cholesterol clearance comes into play. Not only does the liver make cholesterol, it also helps to clear it from the blood!
As you can see in the image above, LDL (and the cholesterol it is carrying) is absorbed through LDL receptors (LDLR) found on the surface of cells. Interestingly saturated fats were shown to decrease LDLR activity in some studies. 2 3
By contrast, a high PUFA diet had the opposite effect and led to increased LDLR levels. 4
This teaches us that problems with LDL are often the result of a lower number of LDL receptors, and saturated fat can impede the function of LDL receptors, causing greater levels of LDL in the blood.
Why all the conflicting information on saturated fat and LDL-C?
The conflicting picture likely arises because of the huge variation in individual responses to eating dietary fat. There are definitely people who are “hyper responders,” meaning they have an elevated reaction to eating saturated fat and cholesterol.
Some people will be at much greater risk for heart disease on a high fat diet. Others won’t be. The trick is to figure out whether you are a hyper responder. For example, cholesterol absorption can vary by individual from as little as 20% to as much as 80% of the cholesterol we eat. 5
This was further evidenced in the recent study by Retterstøl et al which followed healthy adults on a high fat diet and saw variation of between 5 – 107% increase in LDL-C when eating a high fat diet. The average increase was 44% 6

Dietary tips for lowering LDL-C
Here are some good dietary guidelines to consider if your aim is to lower LDL-C.
Rule #1 – Avoid trans fat
Trans fat increases LDL-C in all people. Regardless of genotype, it’s bad for everyone, but avoiding trans fat isn’t as easy as it may sound.
Trans fat can be sneaky. Some food that is labeled as free of trans fat, will actually have small doses of trans fats. Nutrition labels are only required to disclose trans fat if it is found in levels over 0.5 grams per serving. As Harvard Health has reported, all vegetable oils contain trace amounts of trans fat, it’s just the levels are usually low enough that they don’t need to be disclosed.
Despite small amounts of trans fats in vegetable oils, the worst offenders for trans fats are:
- Fried foods
- Processed foods
- Margarine
Rule #2 – Limit saturated fat
As we discuss above, the cholesterol response to saturated fat in the diet is not uniform, it varies by individual.
However, high saturated fat foods will cause increases in LDL-C to dangerous levels in some people. If you see high cholesterol on blood tests, foods like pork, beef, coconut products, cheese, and yogurt will cause an increase in LDL-C, especially for “hyper responders.” To get cholesterol in a range you and your doctor are comfortable with, cut out the saturated fats. We discuss this topic in even greater detail in our blog post about the Bulletproof diet and high LDL.
Rule #3 – Eat healthy omega-3 and polyunsaturated fats
While studies show that saturated fat can reduce cholesterol clearance and increase cholesterol levels, polyunsaturated fats can do just the opposite. 4
Wild salmon, avocado oil for cooking (in moderation), chia seeds and walnuts (in moderation), small amounts of avocado, and algal oil are all good options.
Rule #4 – Cut back on the eggs
A recent analysis of over 600,000 blood samples revealed that ApoE4 carriers are more likely to be hyper-absorbers of cholesterol.
If your doctor has identified you as a hyper-absorber (someone who absorbs more of the cholesterol from food and the cholesterol re-circulating in the body) cutting out high cholesterol foods like eggs and shellfish, as well as adding an absorption blocking drug like Zetia, could help lower LDL-C to healthy levels.
Rule #5 – Experiment with a plant based diet
We are definitely not here to claim that everyone needs to be on a plant based diet all of the time. However, if you’re reading an article about how to lower LDL-C, it’s likely you or someone you know has been told their cholesterol is too high.
Conclusions
As we’ve established in this post, people that find themselves in this situation may have:
- Lower clearance of cholesterol
- Greater synthesis of cholesterol
- Hyper-absorption of cholesterol
In any of these cases, there isn’t as much room to add much more dietary fat into the system because your body is using it to make some bad stuff downstream.
It’s these situations where people can benefit from the type of plant based diets advocated for by the Ornish and Esselstyn camps. Love them or hate them, these diets will bring down LDL-C, we have the studies to demonstrate this.
The problem is these diets are tough to follow over the long term.
Dr. Esselstyn has had some impressive results stopping, and in some cases reversing, advanced heart disease. 7
We hope this post has helped establish that lowering LDL-C is a worthy goal. However, it isn’t the whole picture when it comes to heart health.