TMAO and Heart Disease: Is There Really a Link?

In a previous post (and several others) Kristin covered the ongoing debate about dietary cholesterol, with a particular focus on eggs.
As part of that post, she introduces the molecule called trimethylamine-N-oxide, or “TMAO” for short. TMAO is of great interest in the health community as a New England Journal of Medicine study linked elevated TMAO to heart disease, and eggs are one food that seems to increase TMAO levels.
The TMAO topic is not without controversy.
For example, elevated TMAO is linked to heart disease, but seafood is the biggest source of dietary TMAO and virtually every blue zone (regions where people routinely live to be 100) eats a good amount of fish.
To make matters worse, some of the at-home tests that claim to measure TMAO and its precursors, can be inaccurate. We had this issue when using Viome.
Top Sources
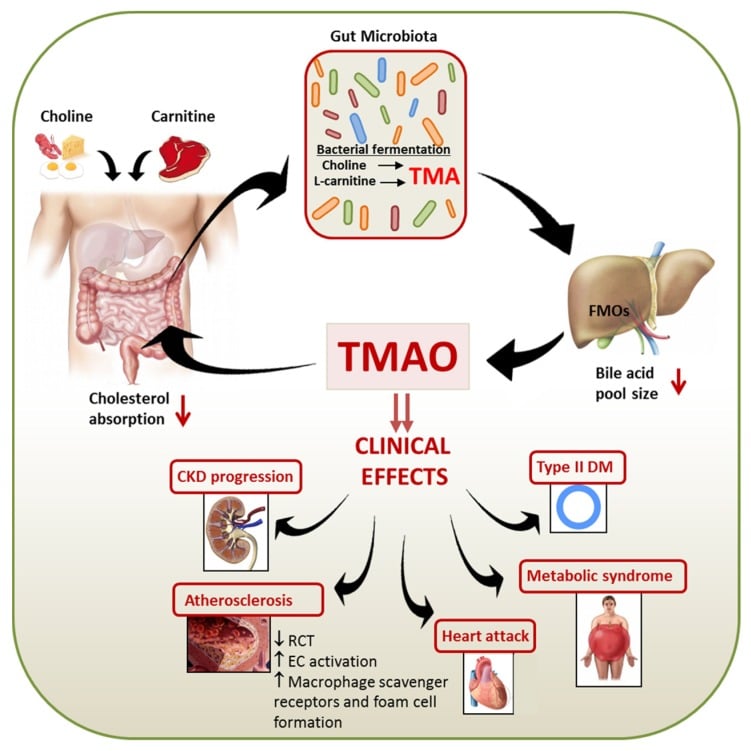
There are two main sources of TMAO, from the diet directly and then via metabolism of other compounds. Targeting dietary sources is relatively straightforward as we know which foods are TMAO rich; however, targeting TMAO synthesis is rather more difficult as several key pathways are involved in its formation.
The major dietary source of TMAO is from dietary choline, saltwater fish and other seafood. TMAO is thought to protect against urea buildup and also mitigate the effects of pressure (useful for fish under the sea, not so useful for us), and interestingly it is the breakdown of TMAO into TMA (trimethylamine) which gives rise to that distinctive seafood smell.
The other major source of TMAO is biosynthesis from trimethylamine (TMA). TMA is a gas generated by bacteria in our gut as they breakdown dietary choline and phosphatidylcholine (think eggs, and dairy). An additional pathway was later described where L-carnitine (think red meats) is converted into γ-butyrobetaine (γBB) and then into TMA again by gut bacteria. Put simply, gut bacteria produce TMAO.
This was confirmed in the NEJM study where researchers gave volunteers broad-spectrum antibiotics which depleted their stomach bacteria. The depleted gut bacteria then correlated with a significant drop in circulating TMAO levels.1 So from a genetic point of view, the obvious target of interest will be the gene that converts TMA into TMAO.
The role of dioxins in TMAO levels
Dioxins are dangerous chemicals, forming part of the “dirty dozen” due to their persistence in the environment and potential to cause great harm.
Typically produced by heavy industry, dioxins are found throughout the world and are readily absorbed into our fat tissues, where they linger for upwards of 5 years. Due to this accumulation in fatty tissue, they tend to accumulate as you move up the food chain… I’m sure you can see where this is going. In a mouse model scientists saw that following a single small dose of dioxin, levels of FMO3 expression and activity were significantly increased, and circulating TMAO was increased 5x.
The authors didn’t perform any long-term exposure studies or follow this administration through to see what if any health impacts the mice developed, but they did report on significant changes in the levels of inflammatory markers and genes which regulate lipid and cholesterol metabolism, all markers of an increased CVD risk. Building on their work, the authors suggest that the dioxin may be targeting the gut microbiota and that this area may be worth investigating therapeutically.
The gut microbiota and TMAO
We know several key digestive processes result in the formation of TMAO including some very common commensal bacteria. What this means is that in otherwise healthy individuals, targeting the gut microbiota in order to reduce TMAO is probably a non-starter. So what about potentially harmful bacteria such as C. difficile and H. pylori?
Well, there is only a single study investigating H. pylori and TMAO, and although the authors show an interesting synergistic effect between the two (TMAO makes the effects of H. pylori infection worse, and vice-a-versa) there is no data to show that one predisposes to the other.1 As a highly novel therapy some researchers have proposed inoculating the gut with commensal bacteria which don’t convert TMA to TMAO such as those from the Methanobacteriales order. These bacteria process TMA into methane rather than TMAO, which while maybe not the most pleasant of gases, is likely to much less harmful than TMAO. However, this concept remains at the hypothetical stage and no work has been performed at the clinical level.
Take home message
The microbiota of the gut clearly plays a big role in determining TMAO levels, as can be seen when it is reduced following antibiotic treatment.
However, as we know the long term health impacts of sustained antibiotic treatment can be just as severe as those associated with TMAO so this isn’t really an ideal therapy. Modulation of the makeup of the gut microbiota is a promising idea, but remains that, an idea.
To me it is clear that TMAO has a role in various diseases when present at high levels. But it is also clear that there is a huge level of variation in people’s responses. We don’t currently have a good handle on what’s causing that variation so the best advice for now would be to check your own levels, and then make a decision based on those in conjunction with your healthcare provider.



All this is very interesting but if FM03’s absence was protective it would be a much better story , no?
Hi there thank you for sharing your thoughts on TMAO. I’ve been reading obsessively about it since my TMAO came back very high, but many of my other heart disease indicators came back low. My very careful GP has imaged my heart, and checked my calcium score too – all A.OK. Hopefully the arterial wall thickness reads out the same. I have read that eating fish will increase TMAO temporarily but the levels dont seem to persist. Have also seen that you can spike TMAO in any population by changing diet to high in choline but vegan’s seem harder to spike because their gut biota populations are different from meat eaters. Since I carry genes that increase my CVD risk – I am trying out a weekday vegan approach and eliminating supplements that contain lecithin and l carnitine since they also increase TMAO. (Hopefully it was my l carnitine containing supplement and mad love of salmon that drove my high initial TMAO result.) Also it seems alcohol results in more TMAO as well (tho resvertrol reduces), so I’ll drop that in January – a good idea anyway. And when I do eat meat once or twice a week i have decided I will be ok with fish – just not in the week before the TMAO retest! Evidence on use of broad spectrum probiotic seems weak but i’m not sure anyone has tested seeding with probiotic and switching to vegan diet to feed the dx biota. we’ll see. will report back on my data point of 1.
What is “a good amount of fish” by scientific standards? If you look at the epidemiology studies done on the blue zones the highest amount of calories consumed from fish is in Sardinia at about 6%. The others get less than 4% of calories from fish. Also some fish contain higher levels of TMAO so that could play a role. I hope I am wrong here because you are the scientist and I am sure you did fact check