The Genetics of Vitamin D Metabolism
Article at a Glance
- Most people don’t realize that the “raw” vitamin D we get through food and supplements is not what our bodies use.
- The body breaks down the “raw” Vitamin D into its active form called calcitriol.
- Several studies have shown that the shorter VDR protein, produced by the presence of the rs2228570 (FokI) ‘C’ allele is more transcriptionally active. This means that given the same levels of vitamin D, someone with the short ‘C’ allele VDR protein will display increased expression of vitamin D-dependent genes. This increased activation of vitamin D-dependent genes results in a greater uptake of calcium from the intestine, which was shown to increase bone density.
- Those with the rs2228570 (FokI) ‘C’ allele may not require vitamin D dietary supplements or at least may require a lower dose, as long as they are obtaining sufficient quantities from exposure to sunlight or through their natural diet.
Genes Mentioned

Contents
You’ve probably read a lot about vitamin D deficiency and its association with poor bone development and health.
After reading this you might have started supplementing your diet with extra vitamin D; but are there those who don’t require this supplementation or for whom it may even be dangerous?
Note: we report on vitamin D genetics in our custom nutrition plans.
Vitamin D 101
- Vitamin D maintains healthy calcium levels in the body by aiding in the absorption of calcium from the intestine.
- It is the combination of both calcium and vitamin D that improves the strength of our bones by increasing bone density. This increased density prevents bone disease such as osteoporosis and rickets.
- Vitamin D is mainly obtained through exposure to sunlight, while small amounts can also be generated from the food we eat or through dietary supplementation.
- The best sources of dietary vitamin D can be found in oily fish, such as salmon or sardines, and many other grocery essentials are now also fortified with vitamin D, such as milk and breakfast cereals.
- Calcium can be found in dairy products such as milk, cheese, and yogurt, as well as spinach and broccoli; again, this nutrient can also be taken as a supplement but can carry significant side effects.
Vitamin D supplements require conversion
- Most people don’t realize that the “raw” vitamin D we get through food and supplements is actually not what our bodies use.
- Ingesting vitamin D is just the start; we then break down the “raw” Vitamin D into its active form called calcitriol. This process of turning vitamin D into calcitrol starts in the liver, and then moves into the kidney, where the calcitriol (1,25(OH)2D) is first made
- Calcitriol, is then utilised in a variety of tissues, mediated via the vitamin D receptor (VDR). So, the VDR gene is important because it is the gateway that unleashes active Vitamin D, calcitrol, into our system.
VDR Genes and Calcium Uptake
So how do calcitrol and VDR control calcium uptake?
Newly converted calcitriol binds to the VDR protein, as encoded for by the VDR gene. It is expressed in most tissues of the body and acts to regulate mineral uptake, particularly that of calcium and iron in the intestine. When calcitriol binds to VDR, the receptor is activated and initiates gene expression of transport proteins such as calbindin. These transport proteins shuttle calcium across the intestinal wall and also hold the calcium in the bloodstream for later use. 1
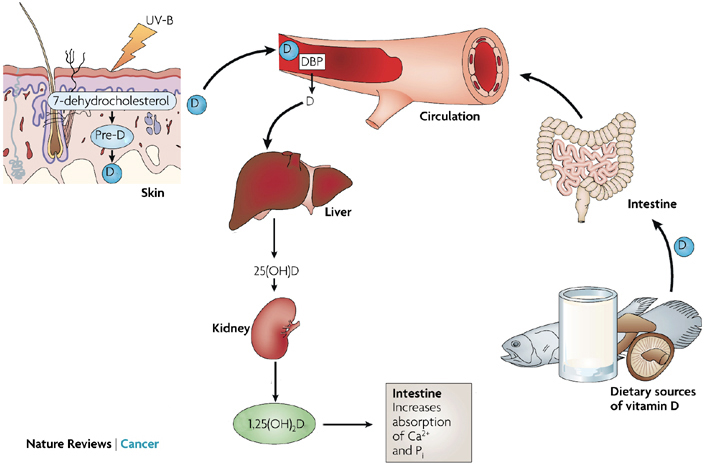
Diagram of Vitamin D activity. 1,25(OH)2D or calcitriol binds with the vitamin D receptor (VDR) protein.
Image adapted from Deeb K, et al., Vitamin D signalling pathways in cancer: potential for anticancer therapeutics. 2007. Nature Reviews Cancer. 7, 684-700.
VDR genes
There are several common polymorphisms within the VDR gene, four of which are rs2228570, rs1544410, rs7975232 and rs731236, traditionally known as FokI, BsmI, ApaI and TaqI respectively.
The BsmI, ApaI and TaqI polymorphisms are strongly associated with one another, that is, the presence of one polymorphism can predict the presence of the others, as they almost always occur together. This is known as linkage disequilibrium.
rs2228570 (FokI)
A ‘start codon’ in the DNA sequence signals where in the DNA sequence the protein chain should be built from. The DNA sequence for a start codon is ATG, which encodes the amino acid methionine. The VDR gene has two potential start codons located very close to each other at the start of the gene.
The SNP rs2228570 (T/C) is located right at the start of the VDR gene and results in the substitution of a methionine (M, Met), encoded by the three-letter codon ATG, for a threonine (T, Thr) amino acid, encoded by ACG at position 1 of the protein (Met1Thr). This removes the first start codon, leading to the second start codon being used to produce a VDR protein.
Therefore, the ‘T’ allele produces a longer VDR protein of 427 amino acids and the ‘C’ allele produces a shorter VDR protein of 424 amino acids. 2 Although of different lengths, both VDR proteins are functional.
rs1544410, rs7975232 and rs731236 (BsmI, ApaI and TaqI)
As the rs1544410 (BsmI), rs7975232 (ApaI) and rs731236 (TaqI) polymorphisms were characterised by researchers in the 1990s, before the sequencing of the human genome, the base changes are often described differently depending on which resource is used.
Therefore these polymorphisms are often referred to by their original names of BsmI, ApaI and TaqI than by their DNA sequence change. Researchers have discovered that the polymorphisms BsmI, ApaI and TaqI, amongst others in the VDR gene, are close enough to one another that they are inherited together more often that would be expected by chance. Therefore, these three variants, and the combination in which they are inherited, are frequently discussed together in the scientific literature.
The ‘G’ allele of BsmI (b), ‘G’ allele of ApaI (a) and ‘T’ allele of TaqI (T) are commonly observed together giving rise to the term baT. The association of other alleles (‘A’ BsmI, ‘T’ ApaI and ‘C’ TaqI) gives rise to the term BAt.
None of the above polymorphisms have an impact on the amino acid produced.
| Reference SNP ID | rs2228570 | rs1544410 | rs7975232 | rs731236 | ||||
| Traditional Name | FokI | BsmI | ApaI | TaqI | ||||
| Allele | T | C (G,A) | G | A | G | T | T | C |
| Traditional Name Variant | f | F | b | B | a | A | T | t |
| Amino Acid | Met | Thr | Non-coding | Non-coding | Ile | Ile |
Summary table of the various different names for VDR polymorphisms and the effect they have on protein coding. For BsmI, AapI and TaqI the alleles which are commonly associated have been coloured giving rise to the baT and BAt terminology which is often seen.
Can we get adequate vitamin D from food?
As discussed above, vitamin D is predominately obtained through exposure to sunlight, with only small amounts being generated from our food. As the modern Western lifestyle has become associated with vitamin D deficiency, due to reduced time spent outside, and typically poorer diets, many people are choosing to supplement their diets with vitamin D.
But does an individuals VDR gene polymorphisms make vitamin D supplementation more or less necessary?
Several studies have shown that the shorter VDR protein, produced by the presence of the rs2228570 (FokI) ‘C’ allele is more transcriptionally active. This means that given the same levels of vitamin D someone with the short ‘C’ allele VDR protein will display increased expression of vitamin D dependent genes (3,4). 3 4 This increased activation of vitamin D dependent genes results in a greater uptake of calcium from the intestine, which was shown to increase bone density. 5 6
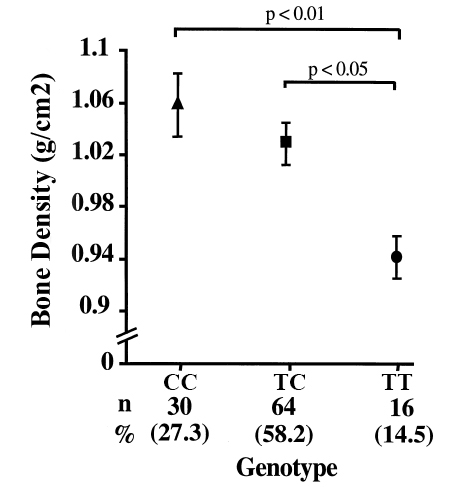
Diagram of rs2228570 (FokI) polymorphisms and their association with bone density. Those with the shorter ‘C’ allele displayed increased bone density due to greater uptake of calcium because of increased VDR activity.
Image adapted from Arai H, et al., A vitamin D receptor gene polymorphism in the translation initiation codon: effect on protein activity and relation to bone mineral density in Japanese women. 1997. Journal of Bone Mineral Research. 12, 915-921.
Similarly, it has been proposed that the complex combinations in which the rs1544410 (BsmI) , rs7975232 (ApaI) and rs731236 (TaqI) are inherited can result in more or less VDR protein being produced, again affecting the rate at which calcium is absorbed from the intestine. However, no consistent effect between studies has been described. 4
VDR Polymorphisms and Heart Health
One possible effect of excessive vitamin D is the onset of heart issues, such as premature ventricular contractions (heart palpitations) or supra-ventricular tachycardia (racing heart), due to the corresponding increase in blood calcium levels.
Muscle relies on a steady supply of calcium ions in order to contract. In the heart this contraction results in the heartbeat; a uniform contraction of the entire muscle which pumps blood around the body. However, if too much calcium is present in the blood, excessive or irregular muscle contractions can occur; in the heart this can manifest itself as an irregular or racing heartbeat. 7
As vitamin D is key in controlling calcium uptake by binding to the VDR protein; and as we’ve seen the rs2228570 (FokI) ‘C’ allele can result in increased VDR, and calcium uptake, activity a key question is:
Are any of VDR gene polymorphisms linked with an increased risk of an irregular or racing heartbeat?
The current answer is we don’t know. Whilst there are anecdotal reports of people with various VDR gene polymorphisms reporting a racing or irregular heartbeat, there are currently no scientific reports confirming this effect.
However, of interest are two studies which investigated the association of VDR gene polymorphisms with coronary heart disease (CHD). In a Chinese population, the rs2228570 (FokI) ‘C’ allele was associated with a reduced risk of CHD,8 several other studies did not observe a similar protective effect. 9 10
Similarly, whilst a study based in the Netherlands described an association between the rs1544410 (BsmI) ‘A’ allele and CHD; a larger study in Germany did not. 11
Take-Home Message
In relation to vitamin D supplementation those with the rs2228570 (FokI) ‘C’ allele may not require vitamin D dietary supplementation or at least may require a lower supplementary dose, as long as they are obtaining sufficient quantities from exposure to sunlight or through their natural diet.
Understanding the impact of the linked rs1544410 (BsmI), rs7975232 (ApaI) and rs731236 (TaqI) polymorphisms is more difficult and further investigation is required to determine a specific recommendation. However, generally vitamin D supplementation is recommended due to the benefits to bone health.
As for cardiac health, whilst there are anecdotal reports of racing heartbeat and heart palpitations in people with known VDR gene polymorphisms, to date there has been no thorough scientific investigation. A role of vitamin D in coronary heart disease, and the association of VDR gene polymorphisms has been widely investigated but results remain inconclusive.



