What is Tetrahydrobiopterin (BH4) and How Can I Make More of it?
Article at a Glance
- Tetrahydrobiopterin (BH4, sometimes THB) is a vital cofactor for numerous enzymes in the body, including those involved in forming nitric oxide (NO), and the key neurotransmitters dopamine, serotonin and epinephrine.
- Additionally, BH4 is the cofactor used by phenylalanine hydroxylase in the conversion of phenylalanine into tyrosine, and its subsequent conversion by tyrosine hydroxylase (TH) into DOPA, the dopamine precursor.
- Low levels of BH4 are associated with several health issues.
- Studies have shown that methylfolate supplementation can increase BH4 levels by preventing its oxidation into BH2 and by supplementing BH4 activity with NOS enzymes further preventing its degradation.
Genes Mentioned

Tetrahydrobiopterin (BH4, sometimes THB) is a vital cofactor for numerous enzymes in the body, including those involved in forming nitric oxide (NO), and the key neurotransmitters dopamine, serotonin and epinephrine. 1
BH4 plays a critical role in both heart and cognitive health. With this important role, many people take supplements that support BH4 levels, and it is frequently discussed together with the one carbon or folate cycle. Before we get into the health impacts of BH4 deficiency and supplementation, and its interactions with the one carbon (folate) cycle, let’s look in more detail how BH4 is formed and how it acts in the body.
BH4 biosynthesis and activity
BH4 is formed from the abundant, simple molecule GTP in a three-step process, each step catalyzed by a different enzyme; GTP cyclohydrolase I (encoded for by the gene GCH1), pyruvoyltetrahydropterin synthase (PTS) and sepiapterin reductase (SPR).2
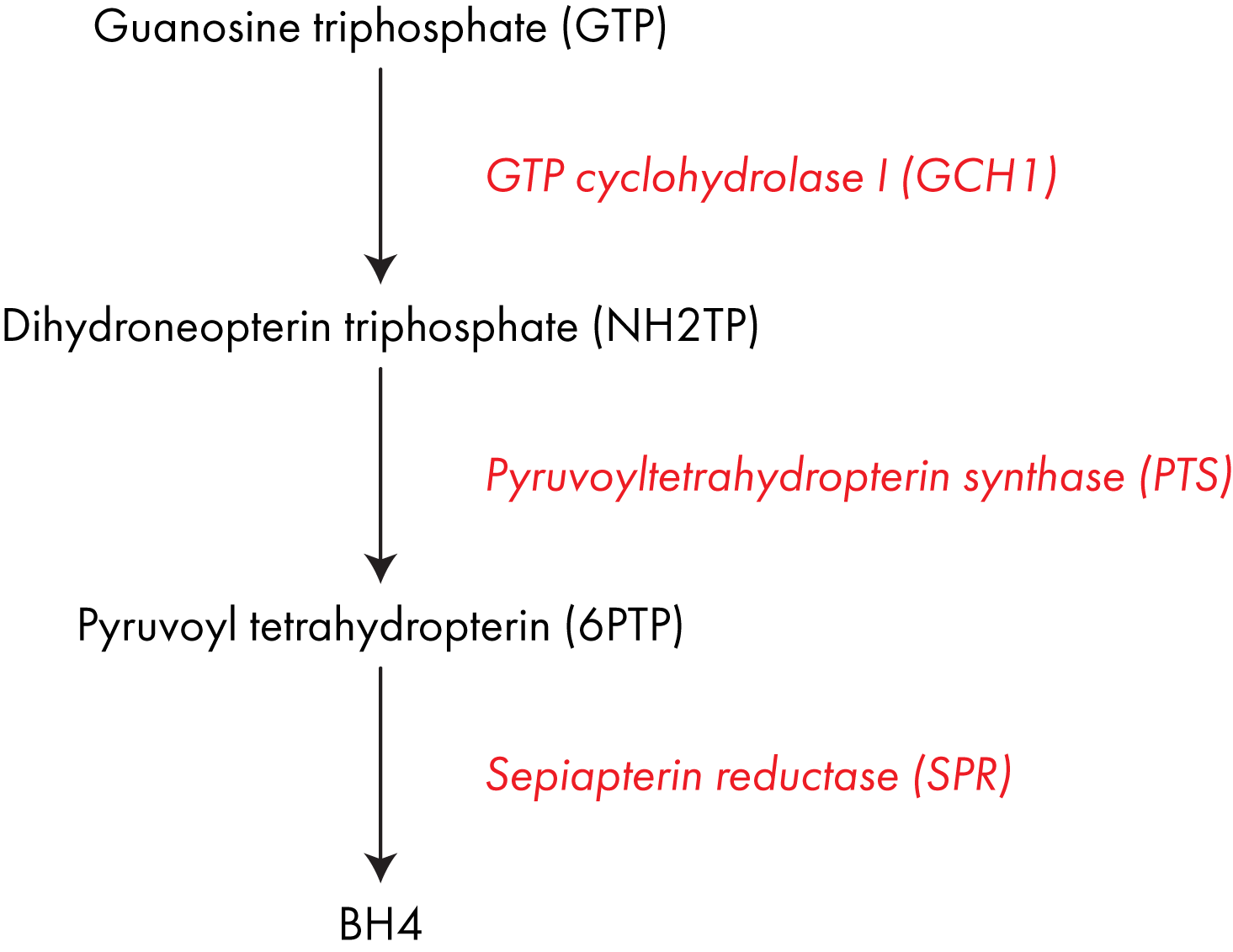
Pathological mutations in any of these genes can lead to BH4 deficiency, it is not yet clear if there are polymorphisms that result in milder reductions in function. Pathway diagram showing the three enzymes involved in converting GTP into BH4.
BH4 as a co-factor
BH4 acts as an important co-factor for many processes in the body.
BH4 helps make nitric oxide
BH4 is an important cofactor for several enzymes including nitric oxide synthase (NOS)1-3 which are responsible for converting arginine into nitric oxide (“NO”). NO is vital in regulating blood pressure in the body. 3
BH4 helps make serotonin
BH4 is also the cofactor for tryptophan hydroxylase (TPH), which converts tryptophan into the neurotransmitter serotonin. You’ve very likely heard of serotonin, it plays an important role in regulating muscle movement and mood. 4
BH4 plays a role in producing dopamine
Additionally, BH4 is the cofactor used by phenylalanine hydroxylase in the conversion of phenylalanine into tyrosine, and its subsequent conversion by tyrosine hydroxylase (TH) into DOPA, the dopamine precursor.5
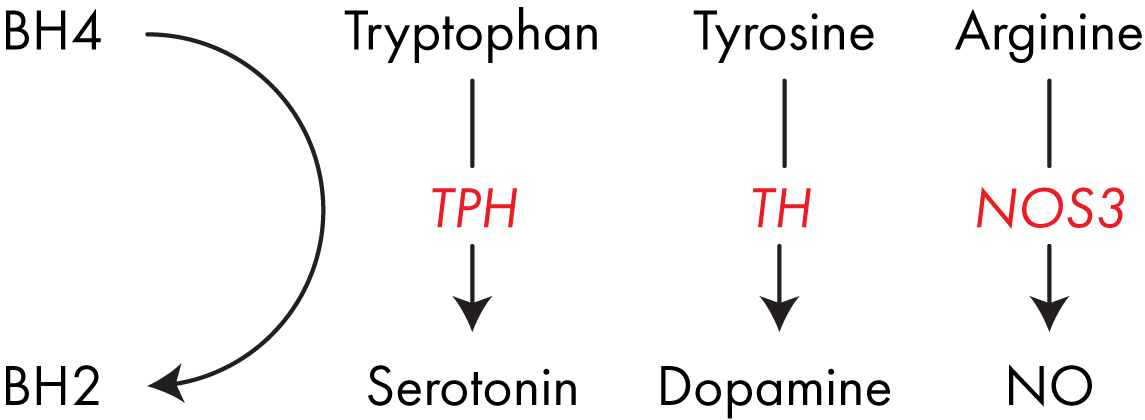
Dopamine is an important neurotransmitter in its own right, but it can also be converted into norepinephrine and epinephrine, the enzymes which control these reactions both use BH4 as a cofactor. 6 BH4 is a key cofactor for several important enzymes involved in neurotransmitter formation and blood pressure regulation.
BH4 recovery
When used as a cofactor in these reactions, BH4 is reduced into a molecule called BH2, or dihydrobiopterin.7
BH4 and NOS uncoupling
BH2 is an interesting product as it can also function as a cofactor for the various NOS enzymes. However, whereas NOS coupled with BH4 converts arginine into NO, NOS coupled with BH2 promotes the formation of superoxides and hydrogen peroxide, two highly toxic compounds, which are associated with several of the symptoms associated with BH4 deficiency. 8
BH2 is converted back to BH4
To avoid production of superoxide, BH2 in the body is rapidly converted back into BH4 through the action of the enzyme dihydropteridine reductase (QPDR).9
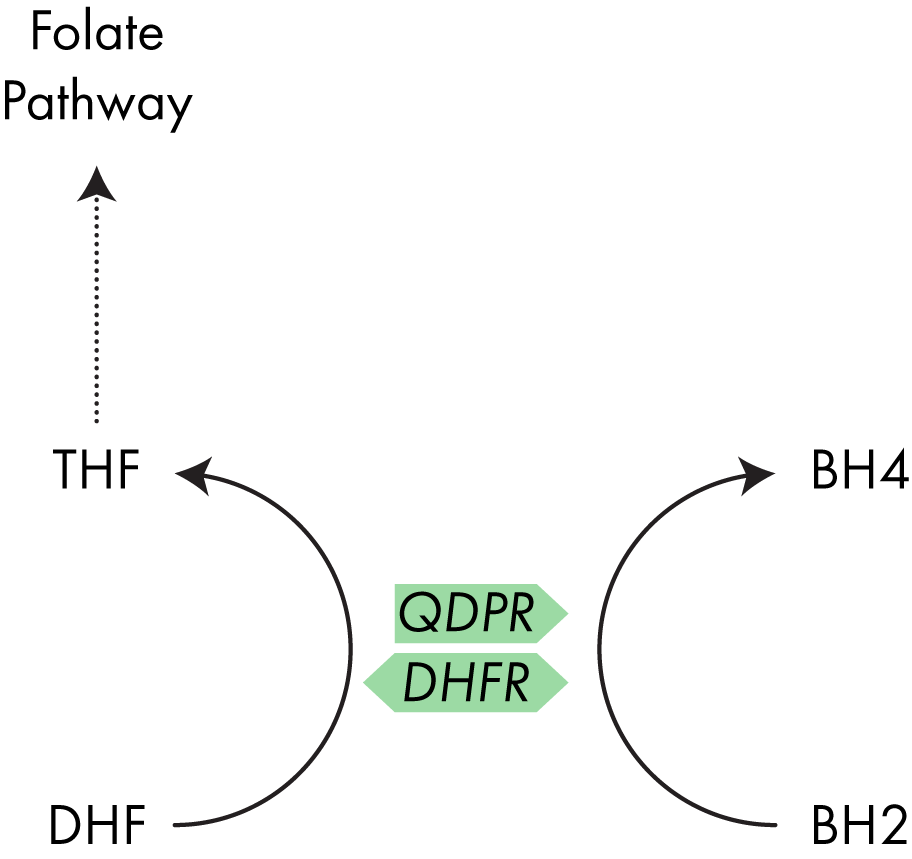
Another enzyme, dihydrofolate reductase (DHFR), which plays a role in the one carbon (folate) cycle, has also demonstrated the ability to convert BH2 to BH4 and I discuss the interaction between QPDR and DHFR below.10 QDPR is the enzyme which typically converts BH2 back into BH4, however the enzyme DHFR has also shown a similar activity.
BH4 Deficiency and Elevated BH2
Extreme cases of BH4 deficiency
Often diagnosed by high levels of phenylalanine (an amino acid which is processed by enzymes using BH4 into tyrosine and eventually dopamine) in the blood, BH4 deficiency is recognized as a severe metabolic disorder which is typically detected early in life due to the occurrence of serious health issues.11 This accumulation leads to impaired intellectual development and loss of muscle control. Furthermore, the lack of BH4 impairs neurotransmitter formation and the production of NO by NOS, which can lead to neurological and cardiovascular issues which John has discussed previously. 12
BH4 and the One Carbon (Folate) Cycle
Right now I bet you’re asking ‘how does this link in with the folate cycle? Well, the answer is quite straightforward, but understanding its relevance to you is a bit more tricky.
- QDPR is the enzyme which is typically responsible for the recovery of BH4 from its reduced form BH2.13
- However, studies have shown that the enzyme DHFR can also perform this recovery activity.
- DHFR, like MTHFR, is an enzyme in the one carbon pathway, hence the similarity in their names. However, their action is rather different. 14
- DHFR sits at the beginning of the folate cycle, MTHFR at the end
- Whereas MTHFR is responsible for converting 5,10-methylenetetrahydrofolate (MeTHF) to 5-methlytetrahydrofolate (MTHF); DHFR converts DHF into THF.19
- THF, a precursor of MeTHF, is often thought as the start point of the one carbon cycle as it is where dietary folate enters the system, and so DHFR activity is key in maintaining cycle activity.
- However, dietary folate is not the only source of DHF; MeTHF can be used in a reaction to form nucleic acids directly, the building blocks of DNA, rather than being used by MTHFR.
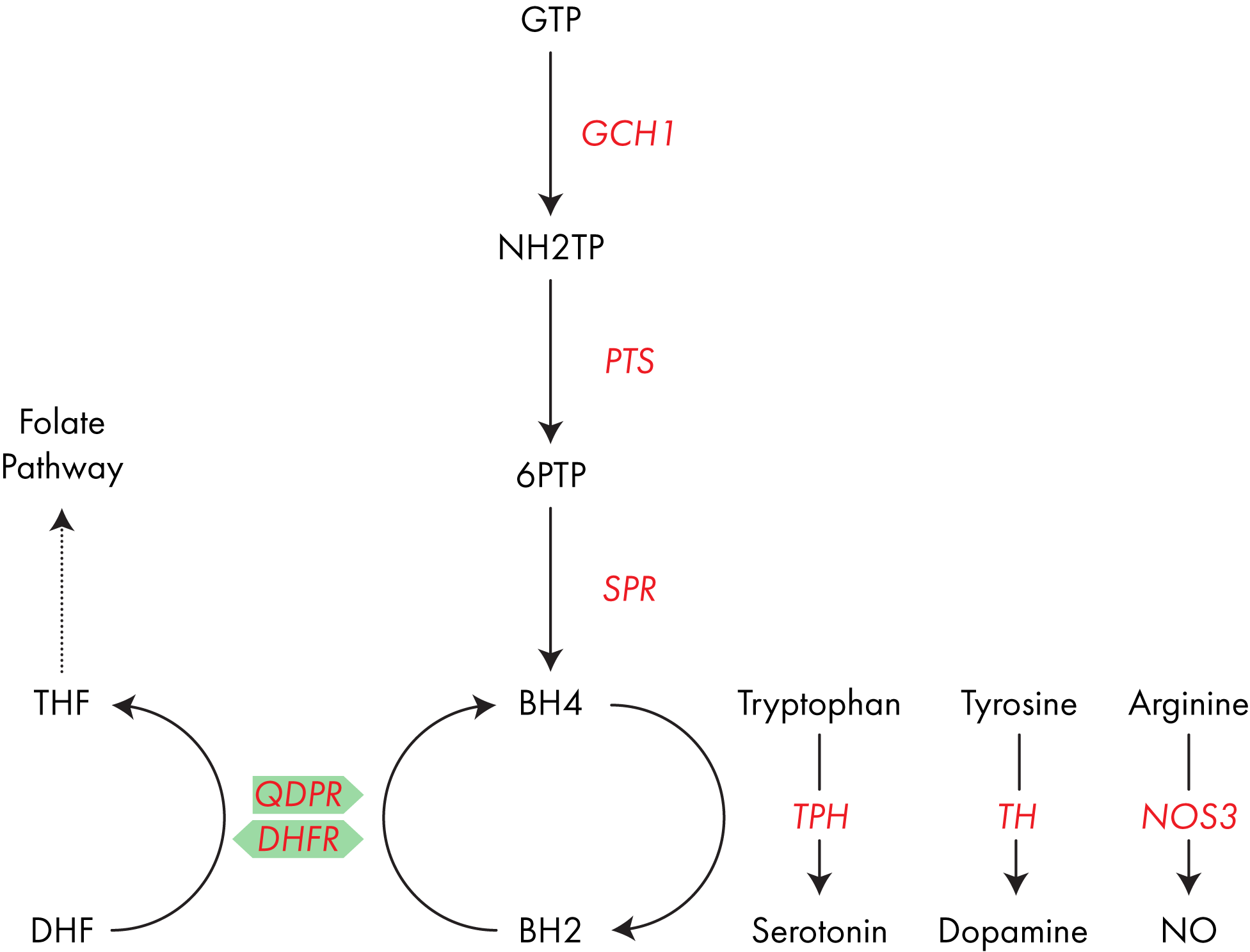
- When used to make DNA, MeTHF is converted into DHF, and so DHFR also plays a recovery role. Understanding how everything links together is quite complex so the pathway diagram below, summarizing everything discussed above, should prove useful.
DHFR can replace QDPR activity
Now we know what the ‘normal’ function of DHFR is, lets have a look at its ‘alternative’ activity. As mentioned above, several studies have shown that DHFR can recycle BH4 from BH2. This study by Xu et al 15 investigates its role in detail, trying to understand how QDPR and DHFR interact. Using mice completely lacking the QDPR gene, the authors discovered that DHFR was able to step in and replace its activity.
However, there are a couple of caveats that go with the study.
Firstly, the affinity of DHFR for BH2 is much lower than for DHF, so the authors propose that the recycling activity of DHFR only kicks in when levels of BH2 are high. Secondly, the authors also state that the affinity of mouse DHFR for BH2 is much higher than that of the human form. So, whilst DHFR may be able to step in to assist BH2 to BH4 processing its importance might be much lower.
Finally, the authors show that high levels of BH2 also lead to a reduction in the availability of THF. The logical explanation for this would be that DHF > THF activity is reduced as DHFR is converting BH2 > BH4. However, this doesn’t seem to be the case. The authors were unable to exactly pinpoint how these were linked but hypothesized that excess BH2 may impact on the activity of other enzymes such as MTHFR.
Folate and BH4 Supplementation Link
The above data provides a link between the two pathways, but is there any evidence that dietary alteration or nutritional supplementation can have a positive effect? If BH2 levels are high then THF levels appear to be reduced.16 However, BH2 levels only reach these severely high levels in the complete absence of QDPR, which will not apply to any readers here. Supplementation with BH4 may increase the levels of BH2 but there is no evidence that this has a negative health effect, or a positive effect on folate activity. Let’s look at it the other way.
Low levels of BH4 are associated with several health issues. Can supplementation with MTHF (the preferred supplementary form) increase BH4 levels? Interestingly, it does seem possible, although the effect is not processed through QDPR or DHFR. Rather, studies have shown that MTHF supplementation can increase BH4 levels by preventing its oxidation into BH2 and by supplementing BH4 activity with NOS enzymes further preventing its degradation.1718
Therefore, MTHF supplementation alone, or in conjunction with BH4, may prove beneficial to those with low levels of BH4.
Closing Thoughts
As always it is important to emphasize that research into these interactions and activities is fairly new. Understanding how various enzymes and pathways interact is a complex study and so whilst beneficial effects may be observed in some, they may not be observed in others.
Direct BH4 supplementation is the most obvious target in those who are BH4 deficient. However, some forum posters report minor adverse effects. In these cases indirect supplementation with MTHF may prove more beneficial in some cases. Readers of this post may also enjoy:
- Our page on Brain and Cognitive Health Genetics
- My post on Telomere length
- My post on the NrF2 Pathway and Nutrition




My compliment to your thorough review here of the 1-Carbon Cycle and BH4/BH2 metabolism. Regarding the cofactors for the various enzymes involved in the 1-C cycle, I know of B2, B9/folate, B6 and B2, however I believe B3/niacin/NA should also be considered a cofactor as NADPH is use in the process. Also, could you elaborate on possible mineral cofactors e.g. Mg, Zn, Cu etc.
PS. I’m not sure why you feel the urge to flag your post as approved medically by a MD, as 95% of MD’s wouldn’t have a clue what you’re taking about here. Their inferior/lacking knowledge on biochemistry and nutrition is known, not surprisingly as the medical curriculum only have 8-12 hours of nutrition or rather deficiency diseases e.g. scurvy, pellagra etc. That’s why Dr Internet is now the new Guru through with people can obtain knowledge, that is not pushed by the Pharmaceutical industry.
Dr. Gardner,
Curious – have you seen any studies on the long-term effects of Kuvan? Our toddler was diagnosed with HyperPhe at birth, and her Phe levels have been steadily increasing. Interestingly enough, after reading some of your articles, we have been supplementing her nutrition with methylfolate and SAM-e (while on an unrestricted diet), but her levels actually went up, not diet. I attributed that to external factors (like, drastic increase of warm weather, being exposed to the sun and the UV rays much more while wearing short sleeves), flu (which causes the body do into a self-canibalization), etc… we are now in the range where Kuvan was prescribed to lower her Phe levels. However, I still want to get to the bottom of this… any insight would be appreciated!!!
Hello Dr Gardner, I stumbled upon your article when I was searching for high phenylalanine and low tyrosine values in serum. My phenylalanine to tyrosine ratio is 3:1 . Due to high phenylalanine I also tested for high Phenyltamine in urine test. I have severe gut issues as well and low serotonin. After reading your article I am guessing it could all be due to a low BH4 deficiency. Any idea how I can increase BH4? I think I may have a mutation in the bh2 to bh4 conversion.
Leah, I have SSA antibody (lupus, rheumatoid, scleroderma, sjogren’s) so staying alive has been a challenge. Given mercuric chloride and chloramphenicol for years as a child until liver failure seemed imminent. I find that it is better to take things away than to add them in. No meds anymore. No dairy (lactose feeds organisms and whey inflammation), only corn, rice and wild rice for grains (others in very small amounts as they also cause inflammation), and no dry beans (worst allergy). Don’t rely on 23 and me-they found virtually nothing. J screen, oat test from Great Plains, Direct Labs and a few other labs out east found the most important mutations. We are still missing a dx on fatty acid oxidation and possibly BH4 since tryptophan and phenylalanine seem to be a problem (5 generations and still undiagnosed). Low Phe/Tryptophan diet has helped that problem but not the others. Keep trying different labs, and not just genetic labs.
Hi, where did you get tested for BH4, if you don’t mind me asking? Thanks!
Dear Aaron,
I have a huge gene report on my daughter and the company closed before I could have anyone interpret, could you help me out? …so I can help fix my daughter.
Thanks,
Tina
Dr. Gardner, Do you do private consultation? I have 23 and Me results, and I think I have a complex profile. I have been having pretty good success but am now running into trouble. I did stop using L Ornithine and Co q10 a while ago when I ran out, I have problems with sulfur and histamine and can tolerate less and less. Face is red, on Prednisone. Was diagnosed w RA and ystemic Lupus about 5 years ago, and the meds initially helped but made me worse. I finally realized after bad reaction to sulfur, (and recalling history) that I had problems w sulfur meds and histamine, which genetic tests seem to confirm. So I stopped using methotrexate, plaquinil (sulfur compounds) and went on regime, which helped tremendously, (when I tried meds again, I had bad reaction) Now in trouble and have appt w Functional medl MD for early July, but please let me know if you do consults. Thank you.
Edward and Robin, Edward, thank you for this . Robin, I have results from 23 and Me, and I can pull up specific genes and see where there are problems. But,did you get yours analyzed.? Does it tell your more information? Where did you go for this. Thank you!
Edit, sorry I meant Aaron.
Hi, what are precursors for BH4? Thanks
Hey Edward GTP is the major precursor as per the diagram below, but there are also several intermediates.
https://www.mygenefood.com/tetrahydrobiopterin-bh4-can-make/#bh4_biosynthesis_and_activity
Sepiapterin runs alongside this pathway but its not clear how big a role this plays.
Importantly BH2 can also be recycled into BH4.
Should have said lab test from HDRI in diagnosing a BH4 deficiency.
Hey Lora, do you have a link to the test, as I can’t seem to find the details online.
Look under, requistion, then undet, vitamins.
http://www.hdri-usa.com/forms/
How accurate is the bh2 bh4 lab test from her I in diagnosing tbed deficiency?
What about normal range phenylalanine but high ratios of tryptophan/LNAA and phenylalanine/tyrsoine? Does this still suggest a bh4 deficiency? Does the MTHFD1 gene variation have a correlation with the deficiency?
Hi,
I have enjoyed the article, thank you. I have gs224 (2 homozygous variants of gch1) according to Prometheus/snpedia. I have many of the Snps listed above along with Nos homozygous Snps. I have been quite sick from something but apparently not from gs224 because my test for phenylalanine / Tyrosine came back normal. Not really sure what to check next, just thought I would leave this info in case it helps someone.
Hi TJ, sorry to hear that you’re feeling sick and that you’ve not been able to hit on what’s causing it, I imagine that’s very frustrating.
Have you tried supplementing with MTHF to see if this helps your BH2/BH4 (Even though you say your phenylalanine/tyrosine were normal)?
Hi Aaron,
Thanks for your response. It’s not celiac’s or an IBD, but Ehlers Danlos, Hashimotos, and mast cell activation that create further challenges for me. I play bit of ‘whack a mole’..make a change in one area and another area dysregulates. My best hypothesis is that perimenopause some how further disrupts BH4 production. I have not directly asked any practitioner to test my phenylalanine levels nor have I asked for a kuvan trial. I would prefer to provide my body with the cofactors to make the BH4 but I don’t seem to be succeeding.
I will use your lovely summary here to discuss additional assessment and treatment options with my functional and pain management practitioners.
My Best to You.
No worries and I’m glad you found my post useful, best of luck with your searching.
Just as a quick note; with your mast cell activation have you considered looking at your histamine, particularly for your AOC1 SNPs?
https://www.mygenefood.com/genes/aoc1/
Hi Aaron,
Yes, I have looked at AOC1, some variation there. But my HNMT variations are more potent. I’m basically heterozygous throughout the gene on the variants 23 and me measured.
Interestingly, blood work found my DAO enzymes to be very low normal, my plasma Histamine to be normal but my plasma tyramine to be high.
My mast cell process is thought to be a primary process. It probably reflects something about the JAK2/Stat3 and IL6 variants I carry.
I’m having some success with L. Plantarum to address the GI issues that reflect my high mast cell load in the lower GI tract.
Warm Regards
Aaron,
How can one directly supplement with BH4? Do you need to get a prescription for Kuvan? If so, how do you communicate this information to a practitioner?
I am someone with GS223 (a GCH1 haplotype),compound mthfr, COMT variants, DHFR variants, CBS variants and even SPR gene variants. I seem unable to tolerate methylfolate. I try to eat 3 cups of leafy greens a day. But this has not prevented me from developing terrible neck and shoulder spasms, as well as other “lovely” symptoms.
Hi Robin,
There are research formulations of BH4 available but I wouldn’t recommend going down that route as there are no requirements for purity etc…
As you say Kuvan is a prescription form, and it may be of benefit to you. Have you discussed this with your practitioner and been rebuffed? If available testing for phenylalanine would be a great step as high levels would be very suggestive of a BH4 deficiency.
Additionally if you feel you’re not tolerating a dietary intake of leafy greens I would definitely recommend mixing it up a little with other vegetables that you can tolerate and consider testing for Crohn’s/celiac disease as this may be impairing your nutrient intake.